Feb. 18, 2017
Bone and joint disorders: Keeping the body's framework healthy
Early diagnosis of arthritis is key to preventing severe disability. Streamlining that diagnostic process reduces patient suffering and provides significant savings to the healthcare system.
Dr. Marvin Fritzler, professor in the Cumming School of Medicine, is a world leader in the diagnosis of autoimmune and inflammatory diseases. He’s developed a series of tests using blood samples and joint (or synovial) fluid that allow physicians to diagnose diseases such as rheumatoid arthritis and osteoarthritis much earlier in their development.
“We have very good evidence that with the earliest treatment, you can have a tremendous impact on the outcome of the disease,” Dr. Fritzler says.
“When patients develop joint pain, more than 100 diseases can present to a doctor. The challenge, after taking a patient history and a physical exam, is to narrow it to less than 10. Then we use diagnostics such as blood tests and diagnostic imaging to identify the specific condition.
“It’s important to differentiate between osteoarthritis and rheumatoid arthritis, because the treatment therapies are as different as the mechanisms of each disease. Biomarkers in blood or joint fluids help to sort out that difference.”
Rheumatoid arthritis (RA) is a chronic autoimmune disorder, where the body’s immune system attacks the body rather than protecting it. Treatment options are much more advanced for RA than they are for osteoarthritis (OA), in part because OA is not just one disease but has three to four different forms and associated treatments.

.
Early diagnosis, and giving the right treatment at the right time, is part of what Dr. Fritzler calls precision medicine. “We’re getting away from one-size-fits-all diagnosis and treatment,” he says. “The treatment of arthritis includes drugs and interventions designed for late disease instead of early disease. An active area of our research is to understand what will work for the treatment of early arthritis.”
"Would you exchange smoking now for a wheelchair at age 60?"
Dr. Fritzler points to some known causes of arthritis. “We know there are a lot of factors that accelerate RA. Smoking for sure. Would you exchange smoking now for a wheelchair at age 60? Also, it’s important to make sure vitamin D levels are appropriate. Certain bacteria in the mouth can cause periodontal disease, and that also affects rheumatoid arthritis.”
Osteoarthritis, on the other hand, can often be inherited or caused by injury, including “wear and tear” OA with chronic inflammation. The treatment may include a specific anti-inflammatory drug that Dr. Fritzler says may work as well for both RA and OA. This research is ongoing.
Engineering principles drive bone scan research
Bone fractures from osteoporosis are more common than heart attacks, breast cancer and stroke combined. “The reality is,” says Steven Boyd, PhD, professor in the Cumming School of Medicine, the Schulich School of Engineering and the Faculty of Kinesiology, “everybody starts losing bone strength as they get older. We don’t know how much bone loss is normal.”
Given that more than 80 per cent of all fractures in people over 50 are caused by osteoporosis, it’s not surprising that Boyd’s research in his role as the Director of the McCaig Institute for Bone and Joint Health focuses on determining who is more likely to break bones, and which bones are more likely to fracture. He says while bone density is often used as a measure of bone health, lots of people with fragility fractures – such as a break you might get after tripping over a rug – have normal bone density. So instead, his research focuses on bone structure, which he believes holds the key to determining who will have the weak bones that are likely to fracture.
“A bone is like the Eiffel Tower,” he says. “The tower is constructed of struts and rods – much like what we see when we examine the internal structure of your bones. That system of struts and rods in a human body is called trabecular structure, and it works on the same principle.”

.
To measure bone structure, Boyd and his team use a high-resolution computed tomography (micro-CT) scanner – one of just 10 in the world – to provide a three-dimensional computer model of the scanned bone. The scanner can identify bone micro-architecture, density and strength information and can be a useful prediction measurement for certain bone and joint disorders – including osteoporosis.
The scanner looks like a big washing machine with a hole in the front where a person puts in a foot or a hand. Due to its size, it scans ankles and wrists, and the occasional knee if a person’s leg is slim enough to fit. It takes just two minutes to scan an area of the wrist or ankle about the thickness of a hockey puck, and the subsequent computer modelling can take up to half an hour after the scan is complete.
The resulting images show a beautiful, intricate latticework of trabeculae – the struts and rods that form our bones’ structure. “We see details of bone structure at the resolution of a human hair. By measuring this bone microarchitecture we can identify weak areas that may be at risk of osteoporosis,” says Boyd.
“The reality is, when bone density decreases, all the trabeculae thin and can actually disconnect some of the structure. While bone density can recover, it rebuilds on the existing bone structure – it doesn’t magically rebuild those disconnected trabeculae. So our hypothesis is that you can recover bone density, but perhaps not the same bone strength.”
The high-resolution CT scanner won’t be a standard medical diagnostic tool in the near future – right now its physical limitations make it only applicable to ankle and wrist. But Boyd sees the role it will play in partnership with other diagnostic tools, allowing researchers – and eventually physicians – even more ways to identify people at risk of bone fractures.
“The key goal is to identify who needs preventive treatment, and how to develop the best treatments possible,” says Boyd.
His team is working on another, related study, this one on the benefits of vitamin D for bone health. “We’re in the second year of a three-year trial about what is the most appropriate dose of vitamin D,” Boyd says. Health Canada approved the study, in which more than 300 people take one of three amounts of vitamin D each day. Vitamin D is sometimes known as the sunshine vitamin, but especially in our northern climate, it may not be possible to get our daily requirement from the sun (we also get vitamin D from certain foods and from supplements).
The role of the muscles in osteoarthritis
“I am interested in muscles,” proclaims Dr. Walter Herzog, PhD.
More precisely, the UCalgary professor in the Faculties of Kinesiology and Veterinary Medicine, the Cumming School of Medicine and the Schulich School of Engineering, is interested in how muscles work, and how they might hold the key to certain medical issues. He’s involved in a variety of research initiatives, including one to do with exercise, diet and childhood osteoarthritis (OA).
“Osteoarthritis is potentially associated with obesity,” says Herzog. “We know that being obese is not good for your joints, leading to earlier onset OA and increased progression of the disease. But what exactly is the cause of OA in people with obesity – is it mechanical loading or is it something else?” (Mechanical loading is the increased stress placed on a knee joint due to extra weight.)
A test group of lab animals was fed a high-fat, high-sugar diet until they reached the weight of a control group of animals. While they all weighed the same, the test group had a much higher percentage of fat – and the results indicate that the ones with the high fat percentage were much more likely to develop OA than those at the same weight but with more muscle.
The study indicates that mechanical loading doesn’t seem to be a factor in causing OA. “We don’t think it’s the weight per se, but the systemic inflammation caused by a high-fat, high-sugar diet that in turn causes the osteoarthritis,” says Herzog. The study also found that exercise combined with a high-fibre diet can reverse the inflammation and could therefore decrease the early onset of OA.
To determine whether adolescents are in similar danger, Herzog’s team the fed the same high-fat, high-sugar diet to adolescent rats and combined it with 12 weeks of exercise five times a week. The animals ran at a leisurely pace for 30 minutes daily on a motor-driven treadmill. More than 80 animals participated in the study.
While the data is not completely analyzed yet, Herzog says preliminary results seem to indicate that joints will improve with a combination of diet and exercise.
.
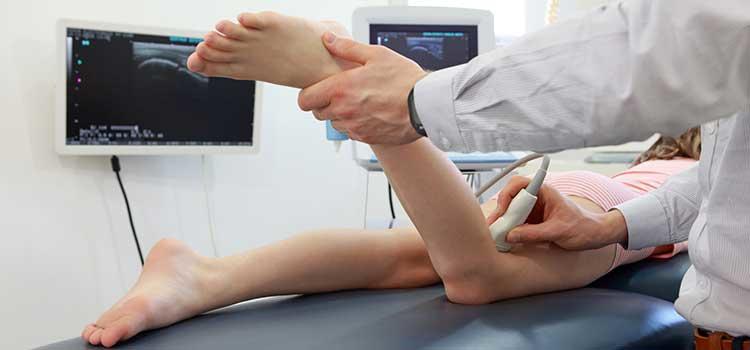
Identifying osteoarthritis from the outside in
Dr. Janet Ronsky, PhD, is looking at knees from the outside in, using advanced technologies to identify osteoarthritis (OA) before traditional x-rays show signs of cartilage wear and tear.
“We know that after a knee injury requiring surgical repair of a ligament, we will see a high rate of osteoarthritis – as much as 40 to 50 per cent,” says Ronsky, who is a professor at the Schulich School of Engineering. “So we’re looking at what starts osteoarthritis – is it mechanical? Is it biological (due to inflammation)? Is it both?”
With an early diagnosis and treatment, it’s possible to mitigate the pain, stiffness and swelling of this chronic condition of the joints. Considering that millions of people around the world are affected with OA, the implications for healthcare management and lifestyle are staggering.
To understand the scope of Ronsky’s work, it’s helpful to first understand the mechanics of OA. She describes it as a breakdown of the biology inside the joint and the surface of the cartilage. Unlike bone, cartilage has no nerves or blood supply, so when that protective cartilage coating erodes, it leaves the nerve- and blood-rich bones to rub together, excruciatingly.
The standard for assessing the severity of osteoarthritis is via clinical x-ray, says Ronsky. “The problem is, you have to already have wear in the cartilage for it to show on that x-ray,” she says.
Her research, then, seeks to learn about early signs of cartilage degeneration based on measuring the mechanical function of the cartilage. In other words, how well does the cartilage support load? Ronsky likens cartilage to a sponge filled with water. A sponge with small holes that is sealed on the top and bottom is like healthy cartilage. When you push down on it, water will flow out slowly and give more resistance to your hand. However, when you push down on a similar size sponge with bigger holes that isn’t sealed, the water flows out faster and it will end up flatter after the same amount of time that you have pushed on it. This is similar to how cartilage with early osteoarthritis behaves. The mechanical marker of cartilage function is the measure of the rate at which the cartilage deforms under the load.
So Ronsky’s research uses a unique combination of state-of-the art medical imaging tools, one of only a few systems worldwide, to see potential issues before there is visible cartilage wear. “We’re looking at how you load the joint when you stand quietly or walk,” she explains. “The changing position of the bones relates to the ability of the cartilage to withstand loading. Within one minute, how does that change over time? When you walk, how do the regions of cartilage contacting throughout your gait cycle relate to your loading patterns?”
Two synchronized video x-rays provide a three-dimensional image of the bones and how their positions change with load or movement over time. They operate at 120 frames per second and have a very high accuracy, better than 0.1 millimetres. A companion MRI shows the 3D shape of the bones and cartilage. Together, they provide a clear picture of cartilage health and lead to the determination of the subject specific mechanical marker for early development of OA. If the cartilage is predicted to be at risk for developing early OA, intervention is possible: a brace to offload the joint, perhaps, or an adjustment in walking pattern to take uneven stress off the joint.
“It’s a pretty exciting tool to help us evaluate this condition and ultimately mitigate the disease progression,” Ronsky says. While it’s still at the research stage, Ronsky is hopeful that in the next five years, this method of assessment will become commercially available.
– – – – –
Explore our programs
– – – – –
ABOUT OUR EXPERTS
Dr. Marvin Fritzler, MD, is a Professor in the Cumming School of Medicine's Department of Medicine with a cross-appointment in the Department of Biochemistry and Molecular Biology. He is also a member of the McCaig Institute for Bone and Joint Health and the Snyder Institute for Chronic Diseases. His research is focused in the area of proteomic biomarkers for precision diagnostics in clinical immunology, autoimmune diseases and arthritis. View Dr. Fritzler's publications
Dr. Steven Boyd, PhD, is a professor in the Cumming School of Medicine, with joint appointments in the Faculty of Kinesiology and the Schulich School of Engineering. He is also the director of the McCaig Institute for Bone and Joint Health. His research is in the area of orthopaedic biomechanics, focusing on adaptive changes to tissues that occur following a joint injury or disease, with particular interest in bone. View his publications
Dr. Walter Herzog, PhD, is a professor in the Faculty of Kinesiology and the Schulich School of Engineering's Department of Mechanical and Manufacturing Engineering. He is also an adjunct professor in Cumming School of Medicine's Department of Surgery and the Faculty of Veterinary Medicine, and a director of the Human Performance Lab. View Walter's publications
Dr. Janet Ronsky, PhD, is a professor in the Schulich School of Engineering's Department of Mechanical and Manufacturing Engineering. She is also an adjunct professor in the Faculty of Kinesiology and the Cumming School of Medicine. Her research group focuses on understanding the links between musculoskeletal system structure, joint mechanics, dynamic joint function, neuromotor control and joint injuries and diseases such as osteoarthritis and scoliosis. View Janet's publications

